Electronegativity is a chemical property that describes the tendency of an atom to attract electrons when it is involved in a chemical bond. It is a measure of how strongly an atom can pull electron density towards itself. The concept was first introduced by the chemist Linus Pauling, and it is typically represented on a relative scale. Electronegativity values can help predict how atoms will interact in compounds.
The Dunathan stereoelectronic hypothesis is a concept in organic chemistry that describes how certain types of orbital interactions can influence the stereochemistry of reactions, particularly those involving the formation or breaking of bonds in organic molecules. This hypothesis was proposed by the chemist D. M. Dunathan in the context of elucidating the mechanisms behind specific stereochemical outcomes observed in reactions.
A four-center two-electron bond is a type of bonding interaction that occurs in certain molecules where a pair of electrons is shared between four atomic centers, rather than the more common two-center two-electron bond found in typical covalent bonds. This concept is particularly relevant in the context of certain types of metal complexes, cluster compounds, and some main-group and transition-metal compounds.
Orbital station-keeping refers to the various maneuvers and methods used to maintain a spacecraft's orbit within desired parameters over time. This is crucial for satellites, space stations, and other payloads in orbit, as their orbits can be influenced by various factors such as gravitational forces from the Earth and other celestial bodies, atmospheric drag (especially for low Earth orbits), and solar radiation pressure.
An intimate ion pair refers to a specific type of ion pair formed in solution, particularly in polar solvents like water. It is characterized by the close association of a cation and an anion that are not fully separated by solvent molecules. In this context, "intimate" indicates that the ions are in close proximity, potentially influencing each other’s properties and reactivity.
Ionic bonding is a type of chemical bond that occurs when atoms transfer electrons from one to another, resulting in the formation of charged particles known as ions. This transfer typically occurs between atoms of significantly different electronegativities, such as metals and non-metals.
A Lewis structure, also known as a Lewis dot structure, is a diagram that depicts the bonding between atoms in a molecule and the lone pairs of electrons that may exist. It was developed by the American chemist Gilbert N. Lewis in 1916. In a Lewis structure: 1. **Atoms** are represented by their chemical symbols (e.g., H for hydrogen, O for oxygen, C for carbon). 2. **Valence electrons** are represented as dots around the atomic symbols.
The term "ligand bond number" isn't standard terminology in chemistry. However, it may relate to the coordination of ligands to a central metal atom in coordination chemistry. In this context, the term "bond number" might refer to the number of bonds that a ligand forms with a central metal atom or ion in a coordination complex.
Linnett double-quartet theory refers to a theoretical model in chemistry that describes the electronic structure of certain types of molecular systems, specifically focusing on the behavior of electrons in larger, complex molecules. While there is limited information available on this specific term, it generally relates to concepts in molecular orbital theory and may involve discussions of resonance, electron coupling, and the stability of certain arrangements of atoms in molecules.
Metallophilic interactions refer to attractive interactions that occur between metal ions or metal-containing species. These interactions can happen due to various factors, including electron sharing, dipole-dipole interactions, and the spatial arrangement of metal centers. Metallophilic interactions are often studied in the context of coordination chemistry, organometallic chemistry, and materials science.
The Morse potential is a mathematical model used to describe the interaction energy between a pair of atoms in a diatomic molecule as a function of their separation distance. It is particularly useful for modeling the behavior of molecular vibrations and is more accurate for describing the potential energy characteristics of bonded systems compared to the simpler harmonic oscillator model.
In chemistry, the term "nascent state" refers to a newly formed species that is in a highly reactive form. This term is often used in the context of nascent hydrogen, which pertains to hydrogen atoms that have just been liberated from a compound and are in a state that makes them very reactive, as opposed to being part of a stable molecule like molecular hydrogen (H₂). The concept of nascent species is important in various chemical reactions and processes.
The Non-Covalent Interactions Index (NCII) is a concept used primarily in the study of molecular interactions, particularly in the fields of chemistry, biochemistry, and molecular biology. While the specific term "Non-Covalent Interactions Index" might not be widely recognized in all scientific literature, the concept generally refers to quantifying or evaluating the strength and nature of non-covalent interactions between molecules.
Dynamic nuclear polarization (DNP) is a technique used in nuclear magnetic resonance (NMR) and electron paramagnetic resonance (EPR) spectroscopy to enhance the sensitivity of these techniques. It involves the transfer of polarization (or alignment) from electron spins to nuclear spins, thereby increasing the observable signal from the nuclei by several orders of magnitude. Here's a brief overview of how DNP works: 1. **Electron Spins:** In the presence of a magnetic field, unpaired electron spins can become polarized.
Electrostatic deflection refers to the phenomenon where an object, often a structural element such as a beam or diaphragm, experiences a change in its position or shape when subjected to an electric field. This principle leverages the forces generated by electrostatic attraction or repulsion between charged elements.
Theoretical chemistry is a branch of chemistry that uses mathematical models and abstractions to explain and predict chemical phenomena. It combines principles from chemistry, physics, and mathematics to provide insights into the behavior of atoms, molecules, and chemical reactions. Key aspects of theoretical chemistry include: 1. **Quantum Chemistry**: This area uses quantum mechanics to study how atoms and molecules interact. It provides a fundamental understanding of electronic structure, bonding, and properties of molecules.
The Chinese multiplication table, often referred to as the "Chinese multiplication chart," is a method used to teach multiplication in a visual and organized way. It is similar to a standard multiplication table but is typically structured differently and may incorporate elements of Chinese numerology or cultural significance. In a Chinese multiplication table, numbers are arranged in a grid format with one set of numbers listed across the top (representing the multiplicands) and another set of numbers listed down the side (representing the multipliers).
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
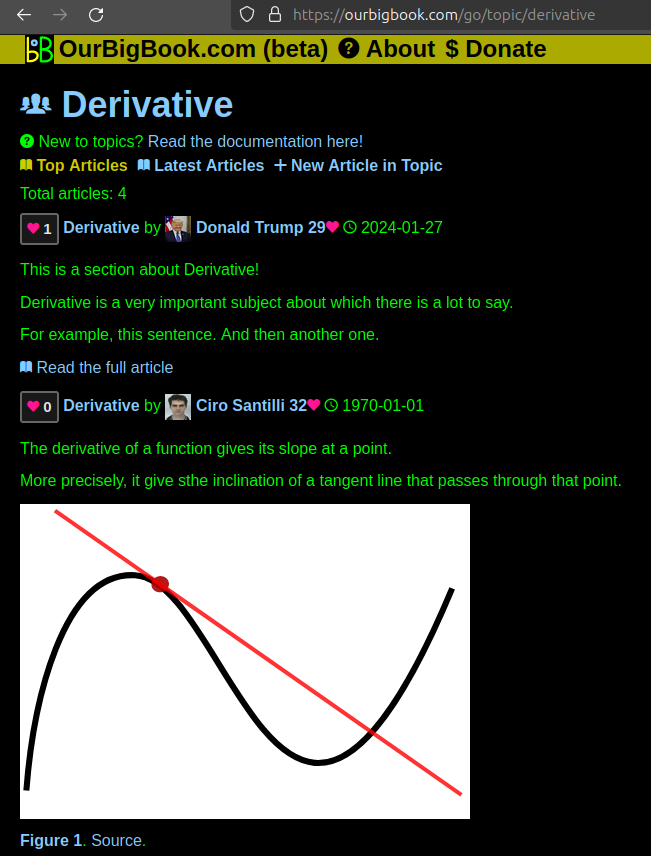
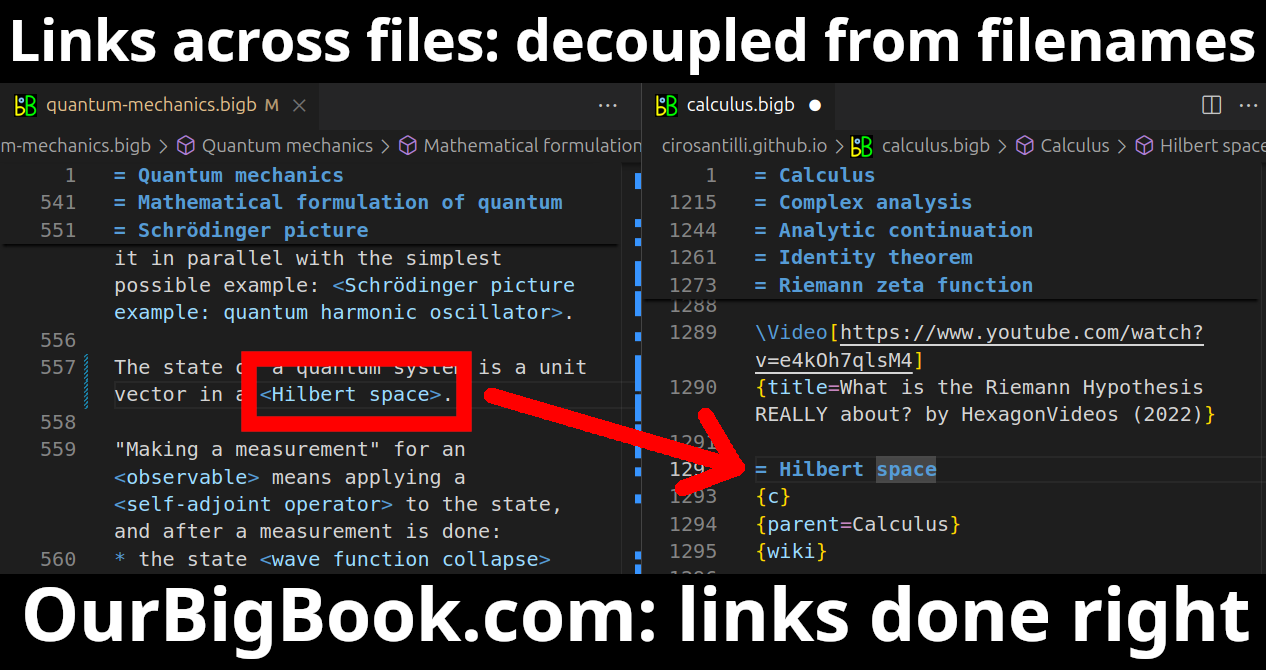
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
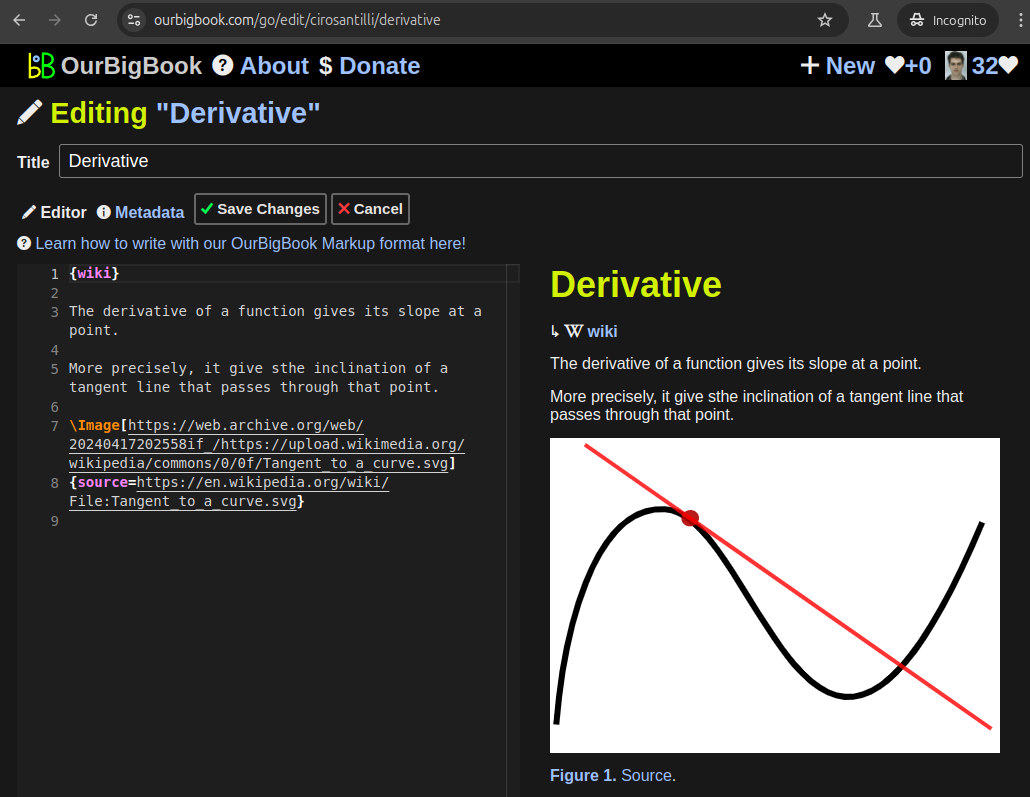
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact