This is closely linked to the Pauli exclusion principle.
What does a particle even mean, right? Especially in quantum field theory, where two electrons are just vibrations of a single electron field.
Another issue is that if we consider magnetism, things only make sense if we add special relativity, since Maxwell's equations require special relativity, so a non approximate solution for this will necessarily require full quantum electrodynamics.
As mentioned at lecture 1 youtube.com/watch?video=H3AFzbrqH68&t=555, relativistic quantum mechanical theories like the Dirac equation and Klein-Gordon equation make no sense for a "single particle": they must imply that particles can pop in out of existence.
Bibliography:
- www.youtube.com/watch?v=Og13-bSF9kA&list=PLDfPUNusx1Eo60qx3Od2KLUL4b7VDPo9F "Advanced quantum theory" by Tobias J. Osborne says that the course will essentially cover multi-particle quantum mechanics!
- physics.stackexchange.com/questions/54854/equivalence-between-qft-and-many-particle-qm "Equivalence between QFT and many-particle QM"
- Course: Quantum Many-Body Physics in Condensed Matter by Luis Gregorio Dias (2020) from course: Quantum Many-Body Physics in Condensed Matter by Luis Gregorio Dias (2020) give a good introduction to non-interacting particles
Allow us to determine with good approximation in a multi-electron atom which electron configuration have more energy. It is a bit like the Aufbau principle, but at a finer resolution.
Note that this is not trivial since there is no explicit solution to the Schrödinger equation for multi-electron atoms like there is for hydrogen.
For example, consider carbon which has electron configuration 1s2 2s2 2p2.
Carbon has electronic structure 1s2 2s2 2p2.
For term symbols we only care about unfilled layers, because in every filled layer the total z angular momentum is 0, as one electron necessarily cancels out each other:
- magnetic quantum number varies from -l to +l, each with z angular momentum to and so each cancels the other out
- spin quantum number is either + or minus half, and so each pair of electron cancels the other out
So in this case, we only care about the 2 electrons in 2p2. Let's list out all possible ways in which the 2p2 electrons can be.
There are 3 p orbitals, with three different magnetic quantum numbers, each representing a different possible z quantum angular momentum.
We are going to distribute 2 electrons with 2 different spins across them. All the possible distributions that don't violate the Pauli exclusion principle are:
m_l +1 0 -1 m_L m_S
u_ u_ __ 1 1
u_ __ u_ 0 1
__ u_ u_ -1 1
d_ d_ __ 1 -1
d_ __ d_ 0 -1
__ d_ d_ -1 -1
u_ d_ __ 1 0
d_ u_ __ 1 0
u_ __ d_ 0 0
d_ __ u_ 0 0
__ u_ d_ -1 0
__ d_ u_ -1 0
ud __ __ 2 0
__ ud __ 0 0
__ __ ud -2 0where:
m_lis , the magnetic quantum number of each electron. Remember that this gives a orbital (non-spin) quantum angular momentum of to each such electronm_Lwith a capital L is the sum of the of each electronm_Swith a capital S is the sum of the spin angular momentum of each electron
For example, on the first line:we have:and so the sum of them has angular momentum . So the value of is 1, we just omit the .
m_l +1 0 -1 m_L m_S
u_ u_ __ 1 1- one electron with , and so angular momentum
- one electron with , and so angular momentum 0
TODO now I don't understand the logic behind the next steps... I understand how to mechanically do them, but what do they mean? Can you determine the term symbol for individual microstates at all? Or do you have to group them to get the answer? Since there are multiple choices in some steps, it appears that you can't assign a specific term symbol to an individual microstate. And it has something to do with the Slater determinant. The previous lecture mentions it: www.youtube.com/watch?v=7_8n1TS-8Y0 more precisely youtu.be/7_8n1TS-8Y0?t=2268 about carbon.
youtu.be/DAgEmLWpYjs?t=2675 mentions that is not allowed because it would imply , which would be a state
uu __ __ which violates the Pauli exclusion principle, and so was not listed on our list of 15 states.He then goes for and mentions:and so that corresponds to states on our list:Note that for some we had a two choices, so we just pick any one of them and tick them off off from the table, which now looks like:
ud __ __ 2 0
u_ d_ __ 1 0
u_ __ d_ 0 0
__ u_ d_ -1 0
__ __ ud -2 0 +1 0 -1 m_L m_S
u_ u_ __ 1 1
u_ __ u_ 0 1
__ u_ u_ -1 1
d_ d_ __ 1 -1
d_ __ d_ 0 -1
__ d_ d_ -1 -1
d_ u_ __ 1 0
d_ __ u_ 0 0
__ d_ u_ -1 0
__ ud __ 0 0Then for the choices are:so we have 9 possibilities for both together. We again verify that 9 such states are left matching those criteria, and tick them off, and so on.
Then you have to understand what each one of those does to the each atomic orbital:
- total angular momentum: determined by the azimuthal quantum number
- angular momentum in one direction ( by convention): determined by the magnetic quantum number
There is an uncertainty principle between the x, y and z angular momentums, we can only measure one of them with certainty at a time. Video 1. "Quantum Mechanics 7a - Angular Momentum I by ViaScience (2013)" justifies this intuitively by mentioning that this is analogous to precession: if you try to measure electrons e.g. with the Zeeman effect the precess on the other directions which you end up modifing.
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
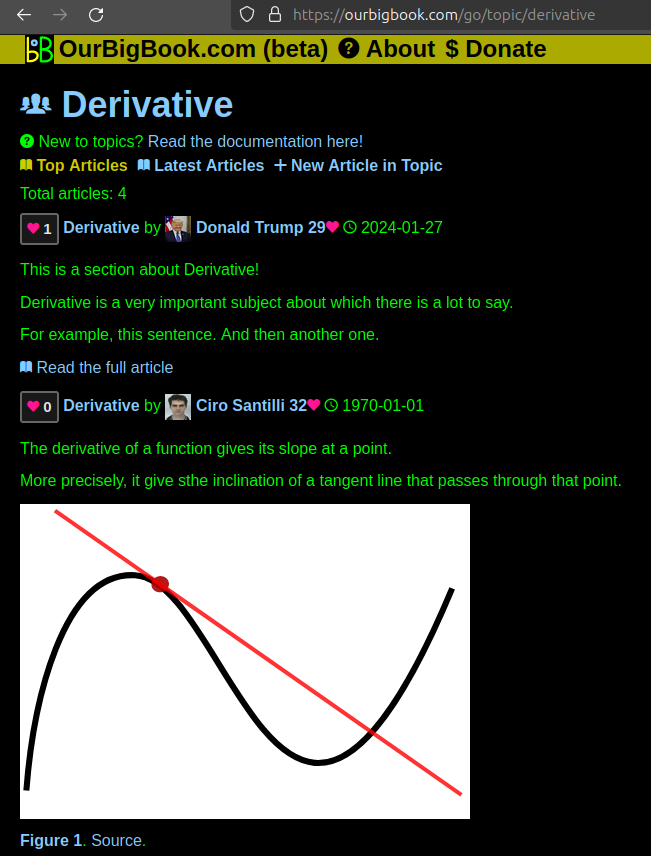
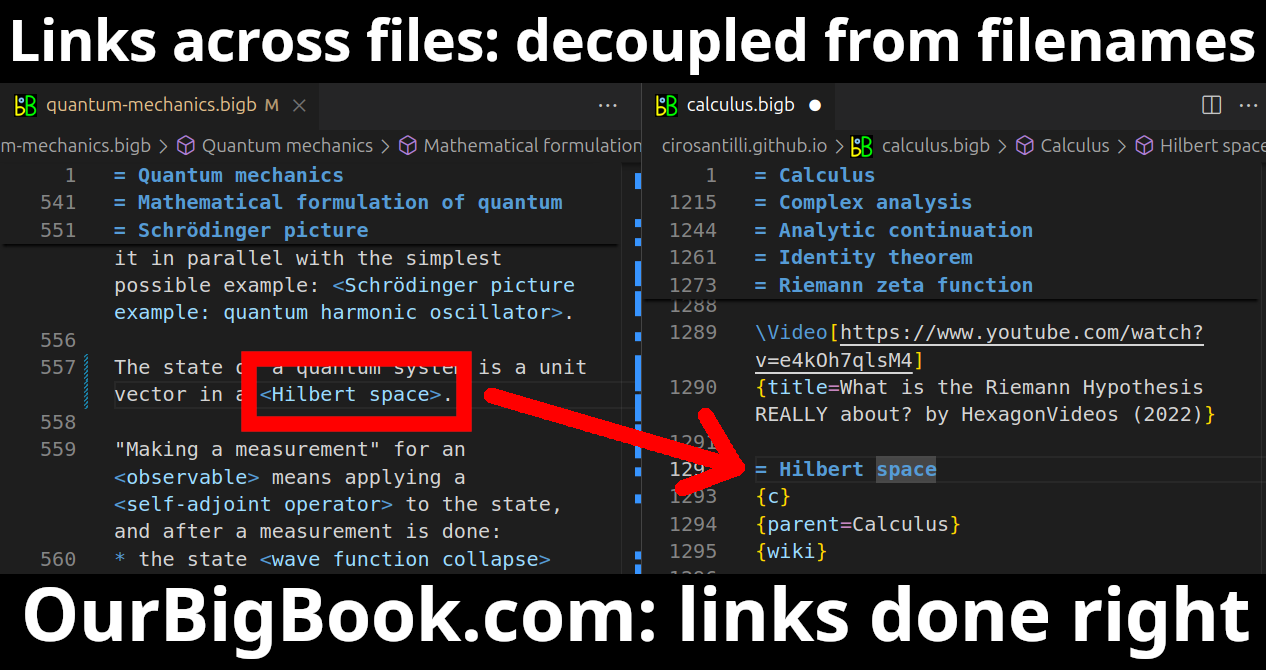
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
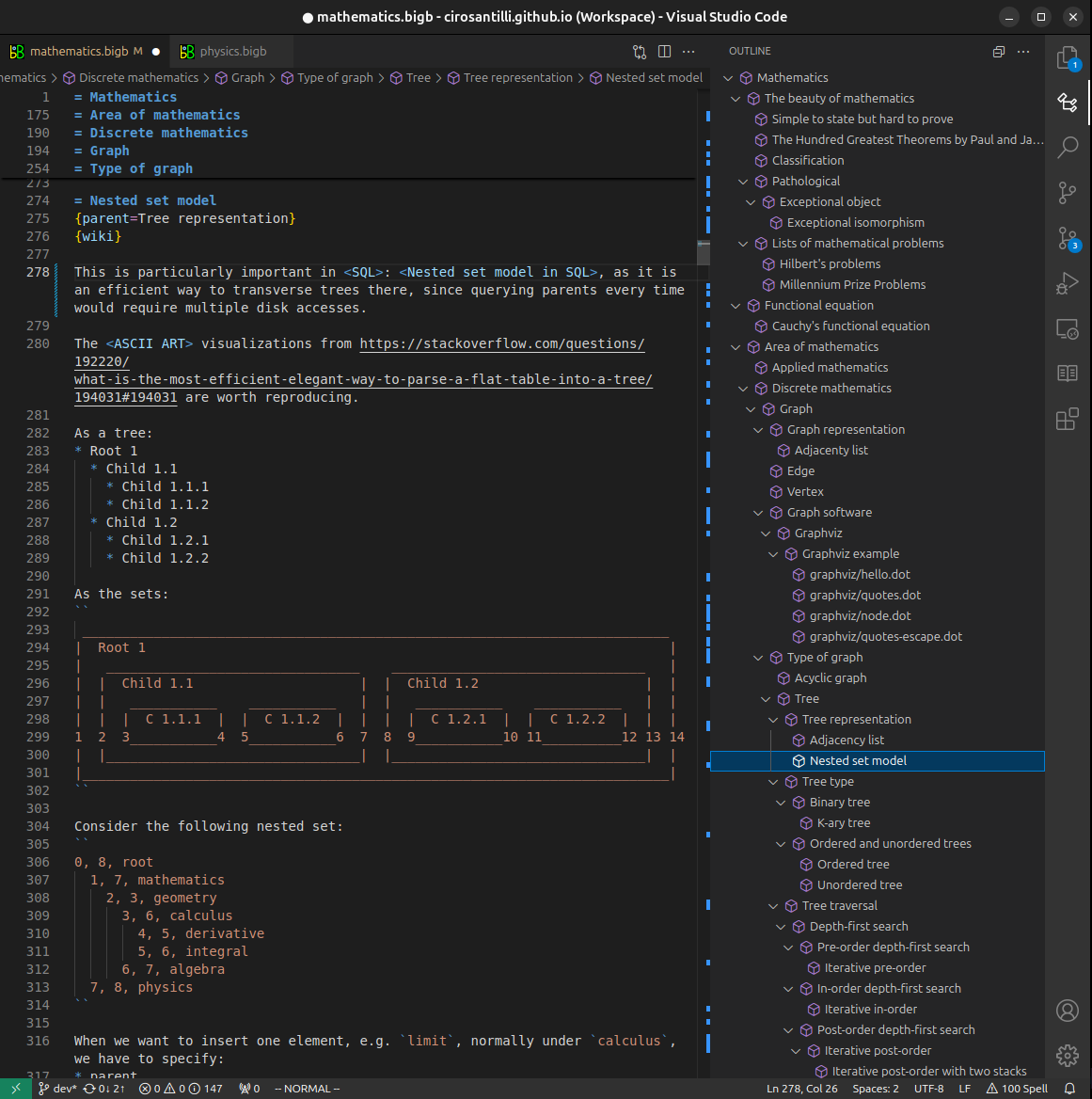
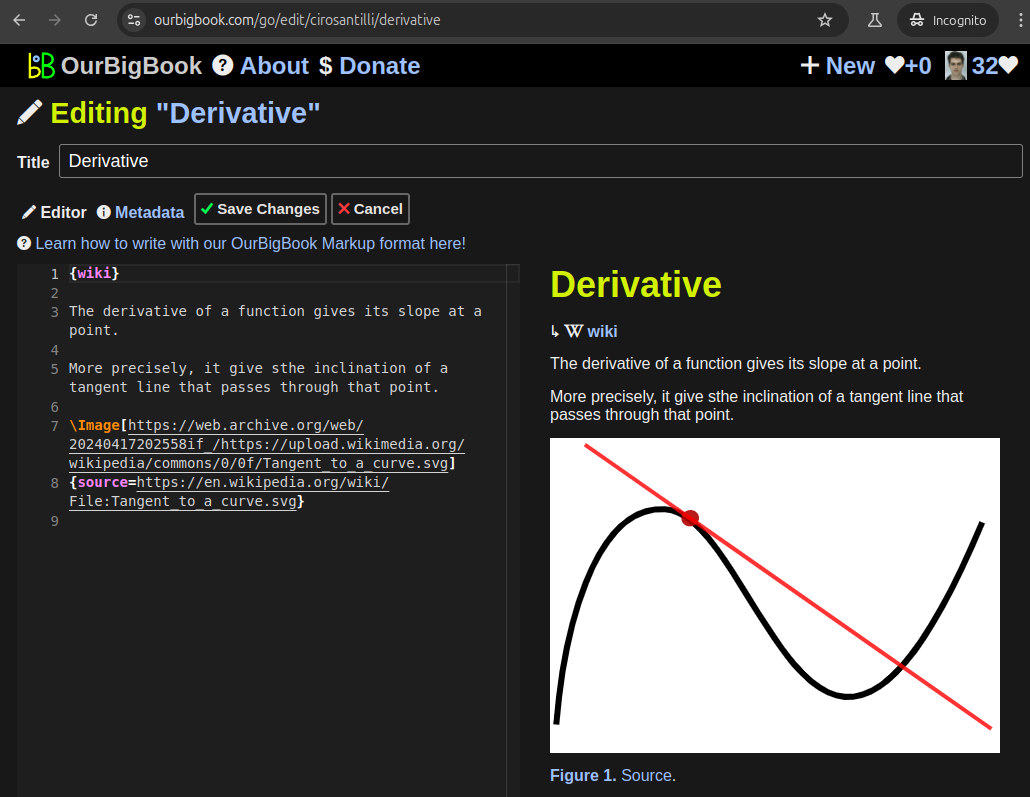
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact