A molecular machine refers to an assembly of molecules that can perform specific tasks or functions at the molecular level, usually through mechanical motion or changes in structure. These machines can carry out tasks such as transporting cargo, catalyzing chemical reactions, or responding to environmental stimuli. Molecular machines are often constructed using components like organic molecules, proteins, or synthetic polymers, and they can be designed to convert energy into motion or perform work in a controlled manner.
The XGC88000 is a model of crawler crane produced by XCMG, a large construction machinery manufacturer based in China. This crane is designed primarily for heavy lifting applications in various sectors such as construction, oil and gas, and infrastructure projects. Key features of the XGC88000 may include: 1. **Lifting Capacity**: It has a significant lifting capacity, capable of handling heavy loads, making it suitable for lifting large precast elements or heavy machinery.
**Obesity** is a medical condition characterized by an excess amount of body fat. It is typically determined using the Body Mass Index (BMI), which is calculated using a person's weight and height. A BMI of 30 or higher is generally considered obese. Obesity can lead to various health issues, including type 2 diabetes, heart disease, certain cancers, sleep apnea, and joint problems. Factors contributing to obesity include genetics, environment, lifestyle choices, and underlying health conditions.
The Varanasi Multi-Modal Terminal is a significant infrastructure project in India aimed at enhancing connectivity and facilitating the movement of goods and passengers through a combination of different transportation modes. Located on the banks of the Ganges River in Varanasi, Uttar Pradesh, the terminal is designed to support riverine transport and provide an efficient interface between inland waterways, roadways, and railways.
Vitreous enamel, also known as porcelain enamel, is a type of glass coating that is fused to a metal surface through a high-temperature firing process. The resultant glassy coating is typically used to enhance the appearance and durability of metal objects, providing a variety of benefits, including: 1. **Aesthetic Appeal**: Vitreous enamel can be produced in a wide range of colors and finishes, allowing for artistic expression and design.
Powder coating on glass refers to a finishing process that applies a protective and decorative coating to glass surfaces using a powder coating technique. Although powder coating is most commonly associated with metals, the process can be adapted for glass as well. Here's an overview of how powder coating on glass works and its benefits: ### Process 1. **Preparation**: The glass surfaces must be thoroughly cleaned and prepped to ensure proper adhesion of the powder coating.
BrickArms is a company that specializes in creating custom accessories and weapons designed for LEGO minifigures and constructions. The company offers a wide range of high-quality, intricately detailed products, including firearms, armor, and other gear, primarily aimed at enhancing play and display options for LEGO enthusiasts and collectors. Founded in 2007, BrickArms operates mainly through online sales and is popular within the adult LEGO community as well as among younger fans looking to expand their LEGO building experiences.
LDraw is a LEGO-oriented computer graphics standard that allows users to create 3D models of LEGO bricks and scenes. It is a file-based format primarily used for generating digital LEGO models, which can be viewed and manipulated in 3D software. The LDraw library contains a vast collection of parts, allowing hobbyists and modelers to replicate or create new designs using digital LEGO elements. LDraw files typically have a ".ldr" or ".mpd" extension. The ".
The FIRST LEGO League Challenge is a robotics competition for elementary and middle school students, organized by FIRST (For Inspiration and Recognition of Science and Technology). The program is designed to engage young people in science, technology, engineering, and mathematics (STEM) through hands-on learning experiences.
Lego Serious Play (LSP) is a facilitated meeting, communication, and problem-solving method designed to enhance innovation and business performance. It utilizes LEGO bricks as a medium for expression and communication, allowing participants to build their ideas, thoughts, and concepts in a tangible form. The method encourages creativity and collaboration among team members. Key aspects of Lego Serious Play include: 1. **Visual Thinking**: Participants construct models that represent their thoughts or ideas, making abstract concepts more concrete.
Ever After High is a multimedia franchise created by Mattel, primarily aimed at children and tweens. Launched in 2013, it centers around a group of teenage characters who are the children of famous fairy tale and folklore characters. These characters grapple with their legacies and the expectations placed upon them by their parents while also seeking to define their own identities. The franchise features a range of products, including dolls, animated web series, books, and a Netflix series.
Furby is an electronic robotic toy that was first released in 1998 by Tiger Electronics. Designed to resemble a small, owl-like creature, Furby can communicate using a unique language called "Furbish," which incorporates various sounds, phrases, and movements. The toy is equipped with sensors that allow it to respond to touch, sound, and light, creating a lifelike interaction.
"Hot Looks" can refer to various concepts depending on the context. Here are a few possible interpretations: 1. **Fashion and Beauty**: In fashion, "hot looks" often refers to trendy styles or popular outfits that are considered fashionable or attractive. This can include specific clothing, hairstyles, accessories, or makeup that are currently in vogue.
Kitty Collier is a unique and creative project that primarily focuses on the art of needle felting, particularly in creating whimsical and imaginative animal figures. The project is run by an artist who shares their work through social media and various platforms, often showcasing detailed, handmade felted animals that are characterized by vibrant colors and intricate designs.
"Britain" generally refers to the island of Great Britain, which comprises three countries: England, Scotland, and Wales. Sometimes, "Britain" is used informally to refer to the entire United Kingdom, which also includes Northern Ireland. The UK is a sovereign country located off the northwestern coast of mainland Europe.
Cox models, commonly referred to as Cox proportional hazards models, are a class of statistical methods used for survival analysis. Named after the statistician David Cox, who introduced them in 1972, these models are particularly useful for studying the time until an event of interest occurs, such as death, disease recurrence, or equipment failure.
Model Railways and Locomotives Magazine is a publication focused on the hobby of model railroading. It typically features a variety of content related to model trains, including features on different scales, tips for building and operating model railway layouts, reviews of locomotives and rolling stock, and articles on techniques for scenery and modeling.
Joshi's Museum of Miniature Railway is a unique and specialized museum located in India, dedicated to the miniature railways. It showcases an impressive collection of model trains and miniature railway setups, featuring intricate details and craftsmanship. The museum offers visitors a chance to explore the history and evolution of railways through these scaled-down replicas. The collection often includes various models and dioramas that illustrate different themes, landscapes, and railway operations, providing insight into the operational and cultural significance of railways.
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
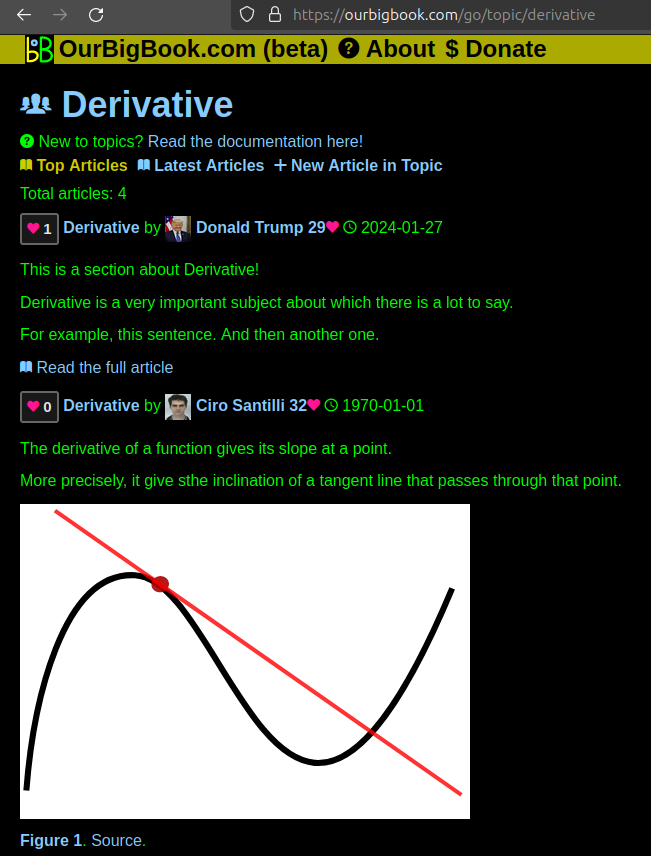
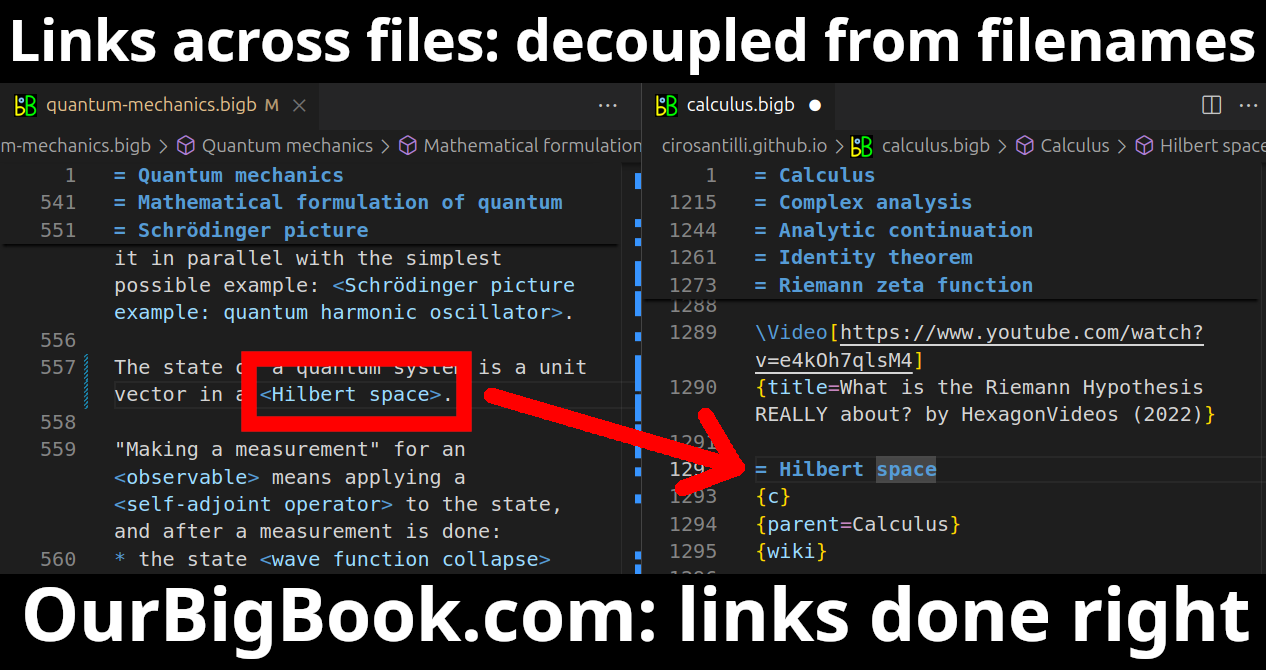
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
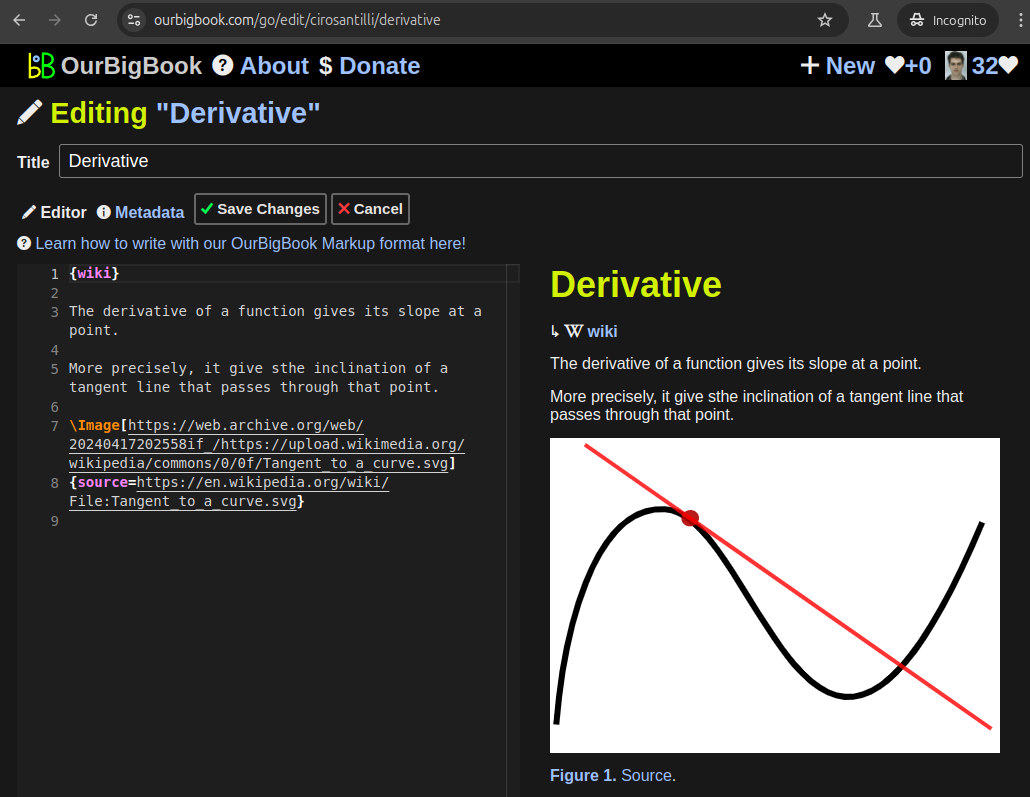
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact