Graymail refers to emails that are not spam but are also not necessarily wanted or needed by the recipient. This category of email typically includes newsletters, promotional emails, and notifications from services or websites that a user has signed up for but may no longer find relevant or engaging. Graymail can clutter inboxes, making it harder for users to find important emails.
A "Joe job" is a term used in the context of online communications and internet marketing to describe a type of spam attack in which an individual or group sends unsolicited emails that appear to come from a reputable source. The goal is often to damage the reputation of the party whose name is being used or to promote a competing service or product by associating it with negative behavior.
An offline reader is a software or application that allows users to access and read content without requiring an active internet connection. These tools are particularly useful for viewing articles, documents, e-books, or other types of media when internet connectivity is limited or unavailable. Here are some key features and aspects of offline readers: 1. **Content Downloading**: Users can download web pages, articles, or documents to their devices while connected to the internet.
Mail-11 is a mail transfer protocol used in the RSX-11 operating system, which was developed by Digital Equipment Corporation (DEC) in the late 20th century. Mail-11 facilitated the exchange of electronic mail between users on the same system or across different systems connected via network links. Key features of Mail-11 include support for the creation, sending, and receiving of messages, as well as the handling of attachments and user directories.
Opt-in email refers to a permission-based email marketing strategy where recipients explicitly consent to receive communication from a sender. This approach is often used by businesses and organizations to build and maintain a list of subscribers who are genuinely interested in their content, products, or services. There are typically two main types of opt-in email: 1. **Single Opt-In:** In this method, a user provides their email address and is immediately added to the mailing list without any further verification.
Microsoft Exchange Hosted Services (EHS) refers to a suite of cloud-based services that provide email filtering, security, archiving, and compliance solutions for organizations using Microsoft Exchange Server. These services are designed to enhance the capabilities of Exchange by protecting it from spam, viruses, and other email-based threats, as well as to provide features for email compliance and data retention.
Mimecast is a cybersecurity company that specializes in email security, archiving, and continuity services. Founded in 2003 and headquartered in London, Mimecast provides a range of services designed to protect organizations from email-based threats, including phishing, malware, and spam, as well as ensuring email continuity during outages. Key offerings from Mimecast typically include: 1. **Email Security**: Protection against phishing attacks, ransomware, and spam. This includes advanced threat detection and response capabilities.
A return receipt is a service provided by postal or courier services that confirms the delivery of a mail item or package. When the sender uses this service, they receive a signed receipt indicating that the item has been delivered and often includes details such as the date and time of delivery. Return receipts can be especially useful for legal documents, important correspondence, or any situation where proof of delivery is necessary. In many cases, return receipts can be requested for various types of mail, including registered or certified mail.
FTP software is a program that allows users to transfer files over the Internet using the File Transfer Protocol (FTP). FTP is a standard network protocol used for the transmission of files between a client and a server on a computer network. Here are some key features and functions of FTP software: 1. **File Uploading and Downloading**: FTP software enables users to upload files from their local machines to a remote server and download files from the server to their local machines.
SaneBox is an email management tool designed to help users organize their email inboxes more efficiently. It utilizes artificial intelligence to sort incoming emails into different folders based on user-defined preferences and past behavior. Key features of SaneBox include: 1. **Smart Filtering**: SaneBox analyzes emails and categorizes them into different folders, such as "SaneLater" for less important emails, helping users focus on what matters most.
As of my last knowledge update in October 2021, Trackbuster does not refer to a widely recognized brand, product, or service. It's possible that it's a new or niche product, service, or company that emerged after that date, or it may refer to a software or tool meant for a specific industry or use case, potentially in areas like logistics, event management, or social media tracking.
Uuencoding (Unix-to-Unix encoding) is a binary-to-text encoding scheme that was commonly used to encode binary files for transmission over protocols that only support text data. Originally developed for Unix systems, uuencoding is designed to convert binary data into a text format that can be sent via email or other text-based systems without loss of data integrity.
X-Face is an experimental email header field that is used in conjunction with the X-Face MIME type to represent a small image or icon associated with the sender's email address. The image is typically sent as a base64 encoded string in the email header, allowing email clients that support the X-Face feature to display the image next to the email.
An FTP bounce attack is a type of network attack that takes advantage of the File Transfer Protocol (FTP) to gain unauthorized access to other systems or to perform denial-of-service attacks. This exploit primarily targets passive FTP connections, where the FTP server allows a client to connect and transfer files.
Gopher clients refer to software applications that allow users to access and interact with content on the Gopher protocol, which was developed in the early 1990s. Gopher is a pre-World Wide Web Internet protocol that provides a hierarchical system for organizing and retrieving information. It structures content in the form of menus and text files, making it relatively straightforward to navigate, but less flexible than the hypertext-based web.
ESPNcricinfo is a comprehensive cricket website that provides news, live scores, statistics, and analysis pertaining to international and domestic cricket. It is one of the most popular cricket websites globally and is known for its extensive coverage of cricket-related content, including articles, features, and player profiles. ESPNcricinfo also offers a range of tools for fans and analysts, such as historical data, player rankings, and match-related statistics.
GopherVR is a platform or framework that focuses on virtual reality (VR) experiences, particularly in the context of education and training. The initiative typically aims to provide immersive learning environments where users can interact with 3D simulations or environments. GopherVR might be used in a variety of fields, such as medical training, engineering, or other areas where experiential learning can enhance understanding and retention of information. Users can engage with content in a more meaningful way compared to traditional learning methods.
The .int domain is a top-level domain (TLD) that is reserved for international organizations established by treaties between governments or other international bodies. It is not as commonly used as other TLDs, but it serves as a way to distinguish official international entities. A list of organizations with .int domain names typically includes well-known international organizations, such as: 1. **NATO (North Atlantic Treaty Organization)** - [nato.int](http://www.nato.int) 2.
Channel Definition Format (CDF) is a file format used primarily for the definition of channels and their attributes in various data-focused applications, particularly in web syndication and for defining content in systems such as RSS feeds and other related formats. CDF allows content providers to specify the structure, content, and metadata associated with the information they are sharing.
Index.dat is a file used by Microsoft Windows, primarily in Internet Explorer, to store various types of data related to browsing activity. Here are some key points about Index.dat: 1. **Types of Data Stored**: Index.dat files can contain information such as URLs of websites visited, cookies, cached webpages, and other browsing history. Depending on the version of Windows and Internet Explorer, multiple Index.dat files may exist on the system, each serving different purposes.
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
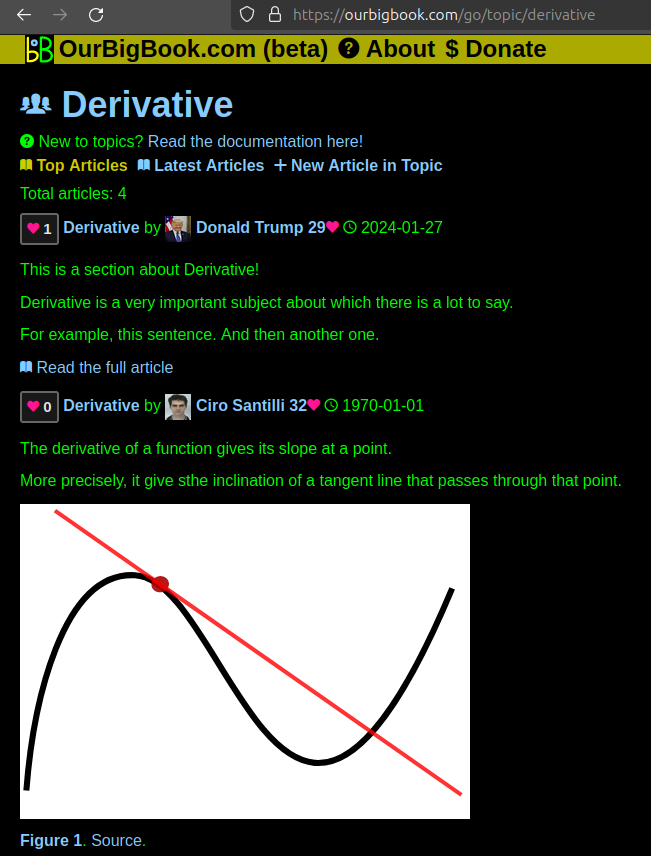
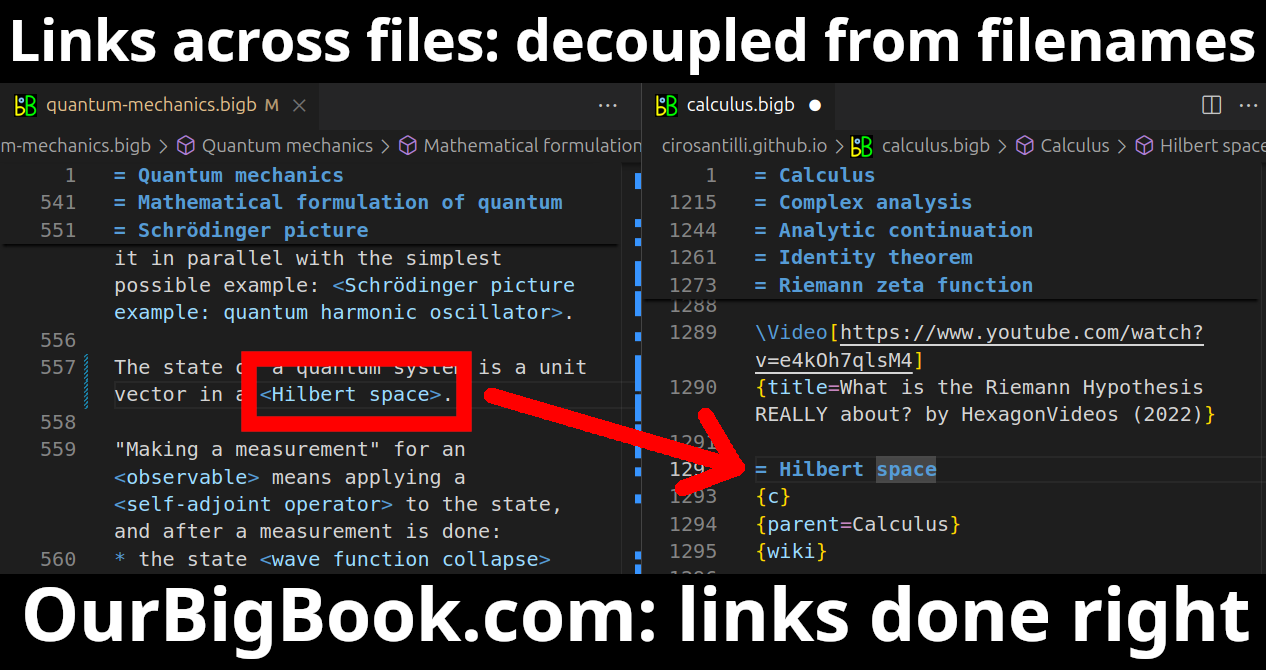
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact