The International Color Consortium (ICC) is an organization founded in 1993 with the goal of achieving greater consistency and accuracy in color representation across different devices and platforms. The ICC develops and promotes color management standards, particularly the ICC Profile specification, which is used to ensure that colors are reproduced consistently across various devices such as monitors, printers, and cameras.
Mango is a vibrant, warm color that is reminiscent of the flesh of a ripe mango fruit. It is typically characterized by a golden yellow to orange hue, sometimes with a slight reddish undertone. This color is often associated with tropical environments, warmth, and energy. In design and fashion, mango can evoke feelings of freshness and excitement and is often used to create a lively and inviting atmosphere.
Persian green is a color often described as a rich, deep shade of teal, incorporating elements of both blue and green. It evokes associations with traditional Persian art and textiles, where vibrant and intricate color palettes are frequently used. This color can be seen in various contexts, including fashion, interior design, and graphic design, and it carries connotations of elegance and sophistication.
Color difference refers to the perceptual or measurable difference between two colors. It can be defined in various contexts, including art, design, photography, physics, and color science. Here are a few key aspects of color difference: 1. **Perceptual Color Difference**: This is how humans perceive the difference between two colors. It can be influenced by various factors, including lighting conditions, surrounding colors, and individual differences in color vision.
Zaffre is a deep blue pigment historically made from cobalt ores. Traditionally, it was created by roasting cobalt arsenate ores, which then produced a blue colorant used in ceramics and glassmaking. Zaffre has been used since the 17th century and is known for its robust and vivid hue. In modern applications, it can refer to cobalt blue pigments that maintain the same characteristic deep blue color.
Achromatopsia is a rare genetic condition characterized by a complete or partial inability to perceive colors, resulting in color blindness. Individuals with achromatopsia typically see the world in shades of gray and have difficulty distinguishing between different hues. The condition is caused by mutations in genes that are important for the functioning of photoreceptor cells in the retina, specifically the cones responsible for color vision.
An anomaloscope is a specialized instrument used to assess color vision, particularly in detecting color deficiencies such as red-green color blindness. It typically consists of a setup that allows the user to match colors using different light sources. The most common type of anomaloscope used in clinical settings has a dial that adjusts the intensity of red and green lights, allowing the test subject to mix these colors to match a standardized yellow light.
The City University test typically refers to the assessments or evaluations that are part of the admissions process for City University of London or other institutions that may similarly use the name "City University." These tests can vary widely depending on the program or course of study for which a student is applying.
Color blindness, or color vision deficiency, is a visual impairment where individuals have difficulty distinguishing certain colors. This condition arises from the absence or malfunction of photoreceptor cells in the retina called cone cells, which are responsible for detecting color. There are different types of color blindness, the most common of which include: 1. **Red-green color blindness**: This includes two main types: - **Protanopia**: Reduced sensitivity to red light due to the absence of red cone cells.
Grapheme-color synesthesia is a neurological condition in which individuals experience a direct and involuntary association between letters and numbers (graphemes) and specific colors. For people with this form of synesthesia, certain characters evoke a consistent perception of colors when they see or think about them. For example, the letter "A" might be perceived as red, while the number "3" could appear green.
The term "Color task" can refer to various activities or assessments depending on the context. Here are a few common interpretations: 1. **Psychological/Neurological Testing**: In psychology, a "color task" might refer to assessments designed to study cognitive processes, such as attention, perception, and processing speed through color-based stimuli.
Congenital red-green color blindness is a hereditary condition that affects an individual's ability to distinguish between red and green hues. It is the most common form of color blindness and primarily results from genetic mutations affecting the photopigments in the cone cells of the retina. **Types of Red-Green Color Blindness:** 1. **Protanopia**: A type of red-green color blindness where individuals have a deficiency in red cone photopigments.
Monochromacy, also known as total color blindness, is a condition in which an individual is unable to perceive colors in the usual way. This can occur due to various reasons, including genetics or damage to the retinal cells responsible for color vision. Individuals with monochromacy typically see the world in shades of gray, as they lack the functional photoreceptor cells that detect wavelengths associated with different colors.
The Purkinje effect, also known as the Purkinje shift, refers to a phenomenon in human vision where the perceived brightness of colors shifts under varying light conditions, particularly in dim or low-light environments. Under bright light conditions (photopic vision), our eyes are more sensitive to longer wavelengths of light, such as yellow and red.
David Wolfe is a mathematician known primarily for his work in the fields of number theory, algebra, and combinatorics. He has made contributions to various mathematical areas, including topics related to modular forms, partitions, and congruences. In addition to his research contributions, he is also recognized for his teaching and mentorship in mathematics. Wolfe may also be involved in mathematical outreach and education, aiming to engage more people with mathematics.
The Criss-cross algorithm is a method used in linear programming to find optimal solutions for certain types of optimization problems, particularly in the context of solving systems of linear equations. The algorithm is especially useful when dealing with problems that involve transportation and assignment structures. ### Key Features of the Criss-cross Algorithm: 1. **Type of Problems**: It is primarily used for transportation problems, assignments, or linear programming problems that can be represented in a tabular format.
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
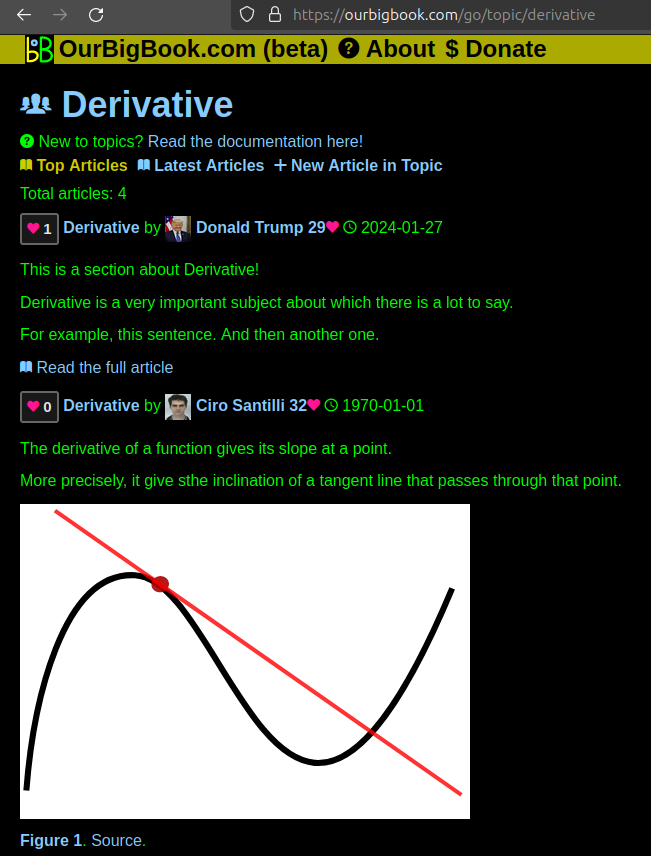
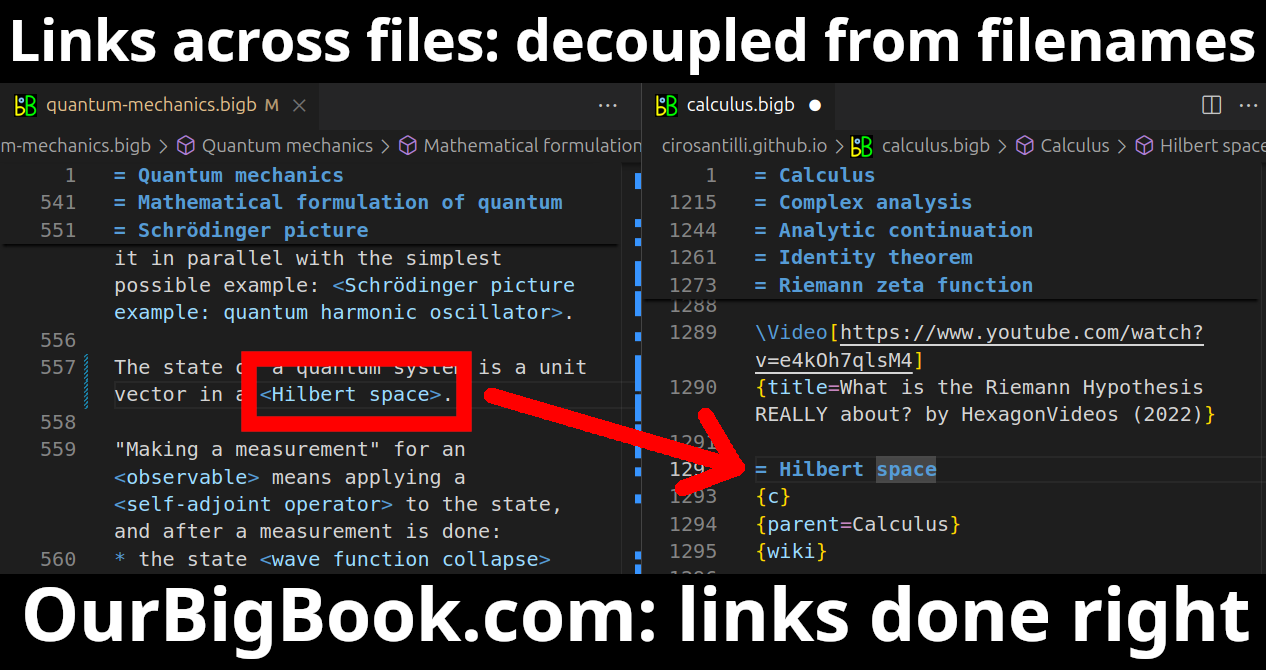
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact