Super Bloch oscillations refer to a phenomenon observed in quantum mechanics, particularly in the context of ultracold atoms and optical lattices. This effect is an extension of the more basic concept of Bloch oscillations, which occur when charged particles, such as electrons, are subjected to an oscillating electric field while in a lattice potential.
Physics in the United Kingdom encompasses the study and application of the fundamental laws of nature, the behavior of matter and energy, and the interactions between various physical systems. The field is explored through various academic institutions, research organizations, and industry, contributing to a wide range of scientific advancements and technological innovations. ### Academic Institutions The UK is home to numerous prestigious universities and research institutions that offer degrees and conduct research in physics.
The Principle of Permanence is a concept that can apply to various fields, including philosophy, science, and law, often referring to the idea that certain states or conditions are enduring and will remain until actively changed.
Mathematical tools refer to a variety of techniques, methods, and structures that can be used to facilitate mathematical reasoning, problem-solving, and analysis across diverse fields. These tools can range from basic concepts and operations to advanced theories and applications. Here are some categories and examples of mathematical tools: 1. **Basic Operations**: Fundamental arithmetic operations (addition, subtraction, multiplication, division) and properties (associative, commutative, distributive).
MinutePhysics is a popular educational YouTube channel created by Henry Reich that specializes in explaining complex physics concepts in a concise and engaging format. The videos typically feature hand-drawn illustrations and animations that help visualize the topics being discussed. Each episode is usually short, lasting around one to two minutes, hence the name "MinutePhysics." The channel covers a wide range of topics in physics, from classical mechanics to modern theories, making complicated ideas more accessible to a general audience.
Actual infinity refers to a concept in mathematics and philosophy that treats infinity as a completed, fully realized entity rather than as a process or a limit. In this context, actual infinity is often contrasted with potential infinity, which represents a process that can continue indefinitely but never actually reaches an infinite value. In mathematics, actual infinity is commonly encountered in set theory. For example: 1. **Set Theory**: The set of natural numbers is considered to be infinitely large.
"A History of Greek Mathematics" generally refers to the study of the development of mathematical concepts, theories, and practices in ancient Greece, which laid significant foundations for modern mathematics. Although there may not be a single definitive text titled "A History of Greek Mathematics," various scholars and historical texts have explored this topic, often focusing on the contributions of key figures such as: 1. **Pythagoras (c.
Algebraic geometry is a branch of mathematics that studies the solutions to polynomial equations through the use of geometric methods. It combines concepts from abstract algebra, particularly commutative algebra, with geometric intuition. Here are some key aspects of algebraic geometry: 1. **Varieties**: The central objects of study in algebraic geometry are algebraic varieties, which are the solutions to systems of polynomial equations.
Pinned article: Introduction to the OurBigBook Project
Welcome to the OurBigBook Project! Our goal is to create the perfect publishing platform for STEM subjects, and get university-level students to write the best free STEM tutorials ever.
Everyone is welcome to create an account and play with the site: ourbigbook.com/go/register. We belive that students themselves can write amazing tutorials, but teachers are welcome too. You can write about anything you want, it doesn't have to be STEM or even educational. Silly test content is very welcome and you won't be penalized in any way. Just keep it legal!
Intro to OurBigBook
. Source. We have two killer features:
- topics: topics group articles by different users with the same title, e.g. here is the topic for the "Fundamental Theorem of Calculus" ourbigbook.com/go/topic/fundamental-theorem-of-calculusArticles of different users are sorted by upvote within each article page. This feature is a bit like:
- a Wikipedia where each user can have their own version of each article
- a Q&A website like Stack Overflow, where multiple people can give their views on a given topic, and the best ones are sorted by upvote. Except you don't need to wait for someone to ask first, and any topic goes, no matter how narrow or broad
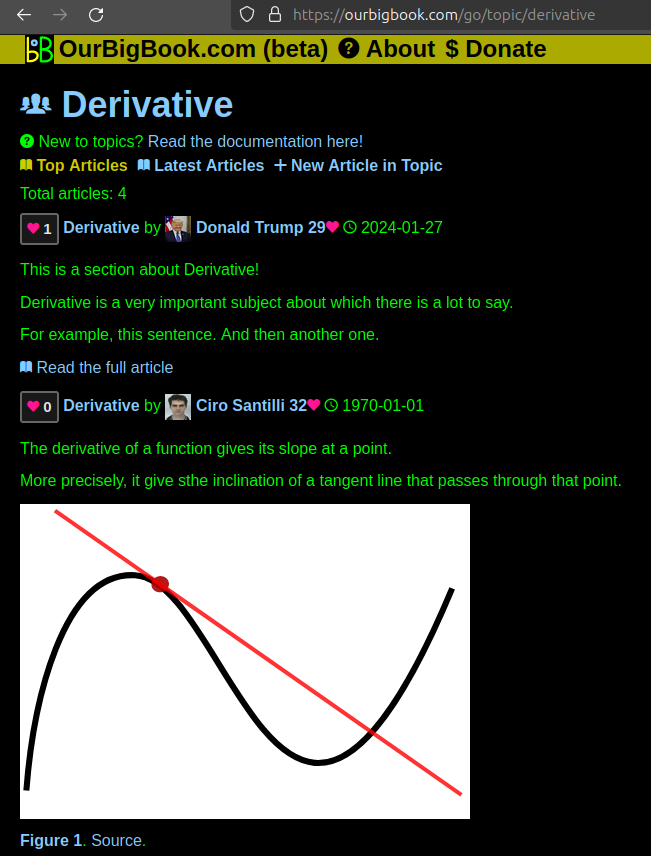
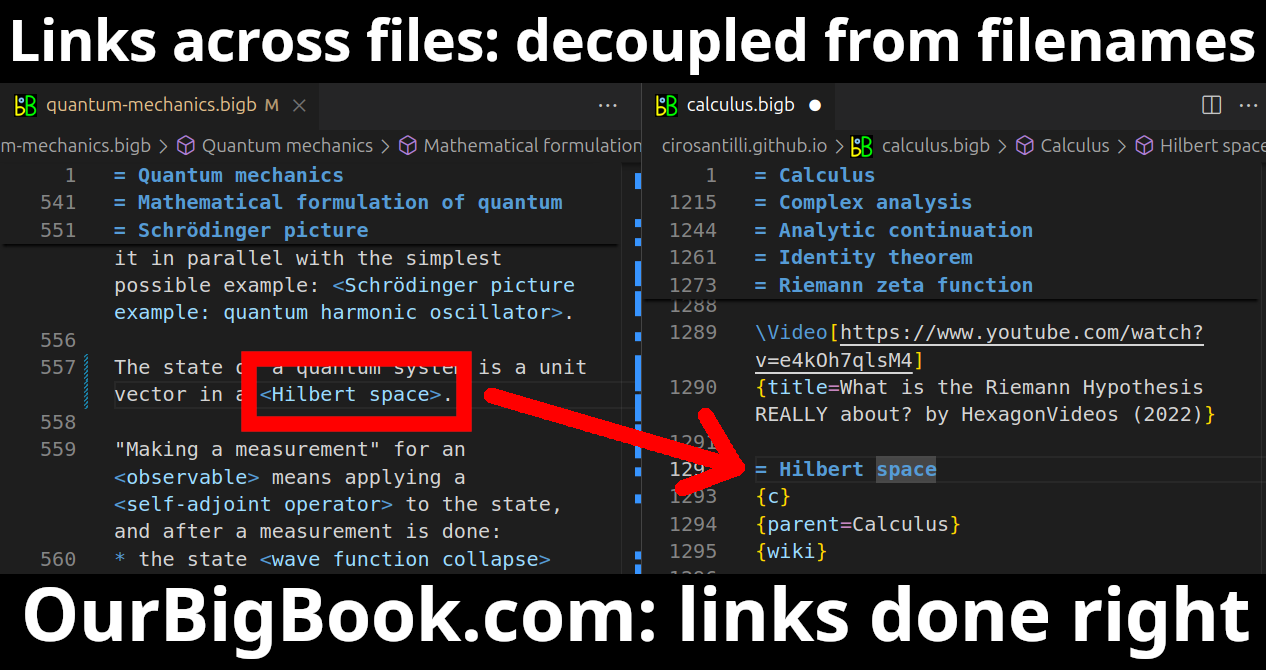
This feature makes it possible for readers to find better explanations of any topic created by other writers. And it allows writers to create an explanation in a place that readers might actually find it.Figure 1. Screenshot of the "Derivative" topic page. View it live at: ourbigbook.com/go/topic/derivativeVideo 2. OurBigBook Web topics demo. Source. - local editing: you can store all your personal knowledge base content locally in a plaintext markup format that can be edited locally and published either:This way you can be sure that even if OurBigBook.com were to go down one day (which we have no plans to do as it is quite cheap to host!), your content will still be perfectly readable as a static site.
- to OurBigBook.com to get awesome multi-user features like topics and likes
- as HTML files to a static website, which you can host yourself for free on many external providers like GitHub Pages, and remain in full control
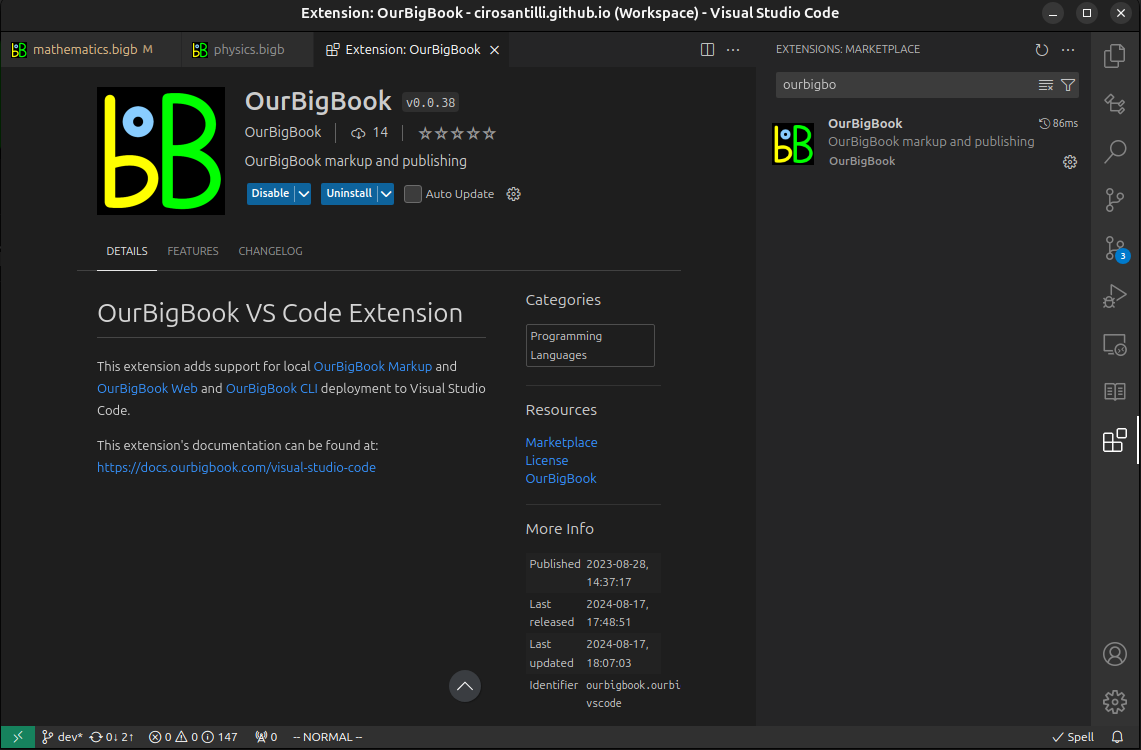
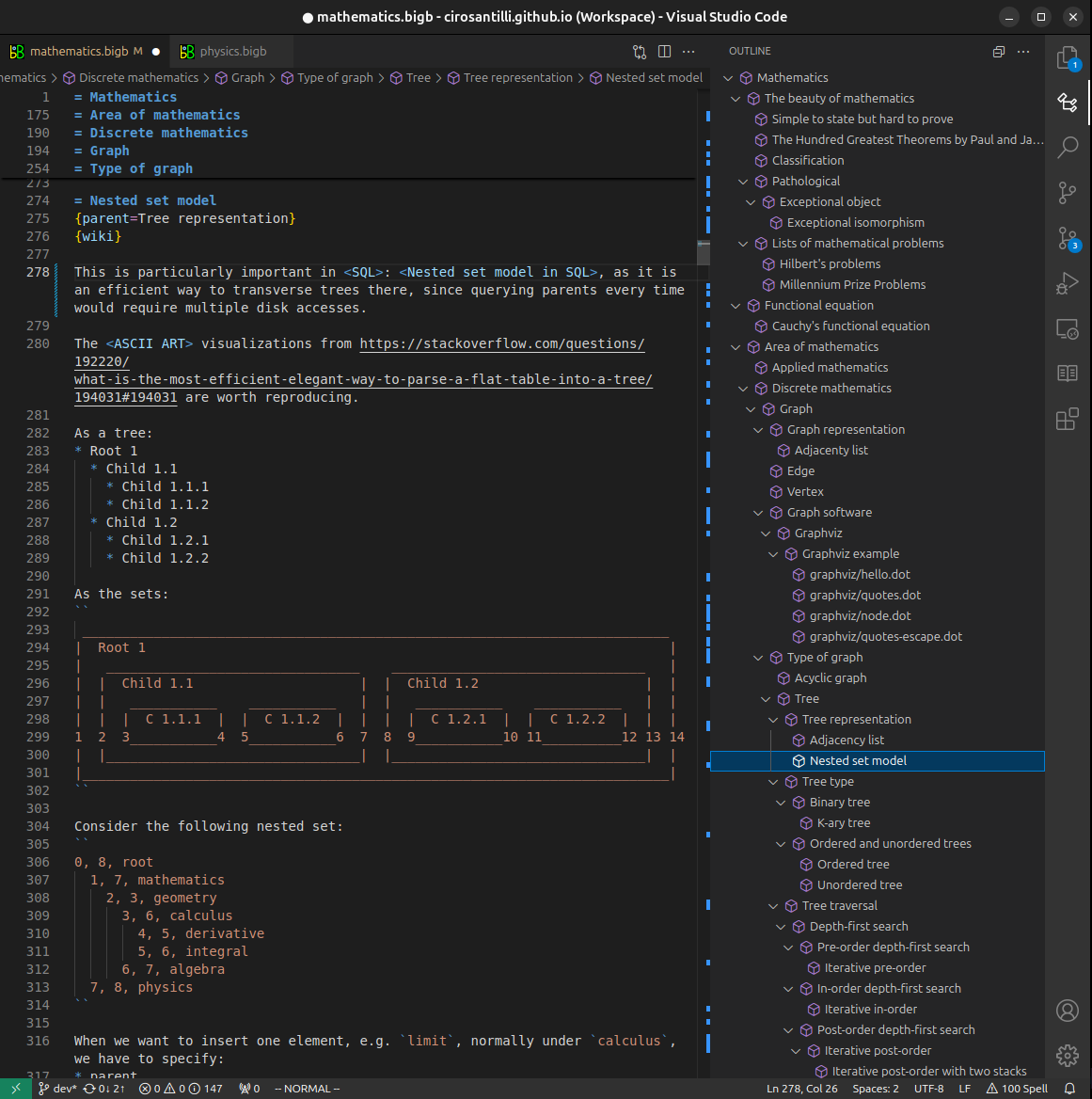
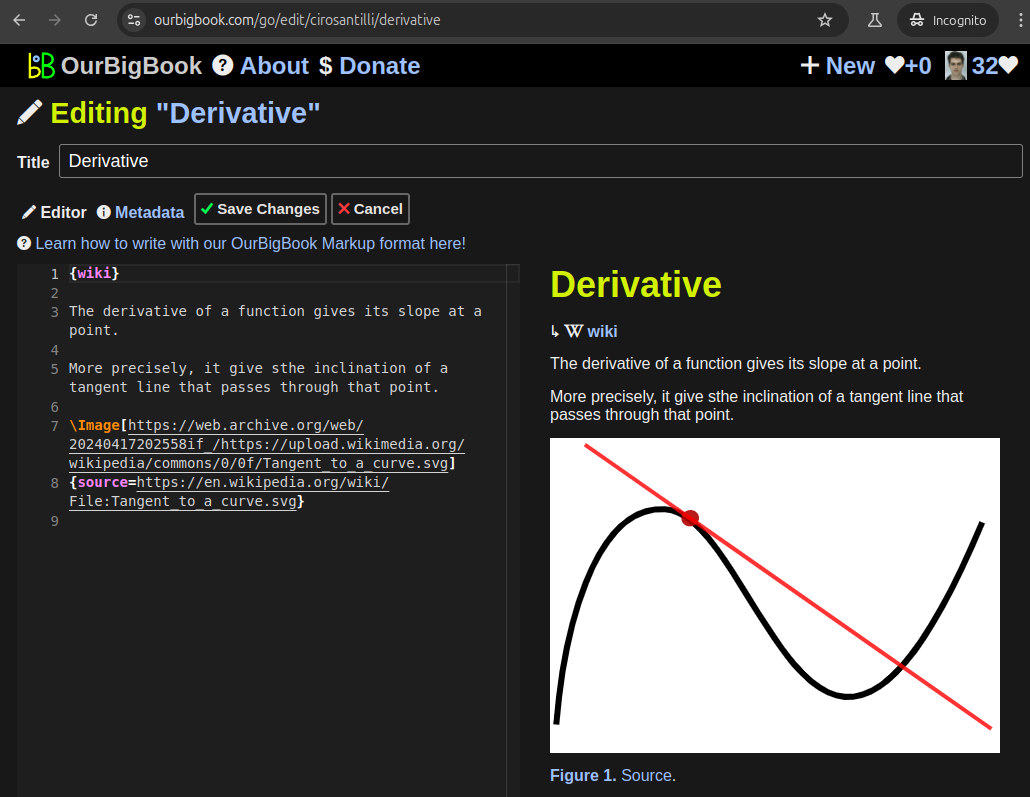
Figure 3. Visual Studio Code extension installation.Figure 4. Visual Studio Code extension tree navigation.Figure 5. Web editor. You can also edit articles on the Web editor without installing anything locally.Video 3. Edit locally and publish demo. Source. This shows editing OurBigBook Markup and publishing it using the Visual Studio Code extension.Video 4. OurBigBook Visual Studio Code extension editing and navigation demo. Source. - Infinitely deep tables of contents:
All our software is open source and hosted at: github.com/ourbigbook/ourbigbook
Further documentation can be found at: docs.ourbigbook.com
Feel free to reach our to us for any help or suggestions: docs.ourbigbook.com/#contact